
An investigational noninvasive, pain-free test for A1C and glucose


ELG3 Corporation is in the investigative phases for a diabetes category game changer that has the potential to rival standard methods of glucose testing with a U.S. market size of about 30 million patients. For this study, we are looking for 100 patients, ages 25-65, who are relatively stable and with glucose levels less than 300 mg/dl.
The Easy Light Glycometer (ELG) is the first product designed specifically for patients with type 1 and 2 diabetes mellitus that is in a clinical trial to measure both A1C and glucose, without the pain and inconvenience of a fingerstick or inserted device. The ELG sensor system uses pulse photometrics to measure blood glucose concentrations.
Unlike current OTC finger sticks and under-the-skin CGMS, this device does not puncture or even scratch the skin. Through the investigation, we hope to demonstrate that the ELG device delivers the same level of >95% on both precision and accuracy as compared to standard whole blood assays for A1C. We hope to enroll a diverse diabetes patient profile to establish comparable precision and accuracy.
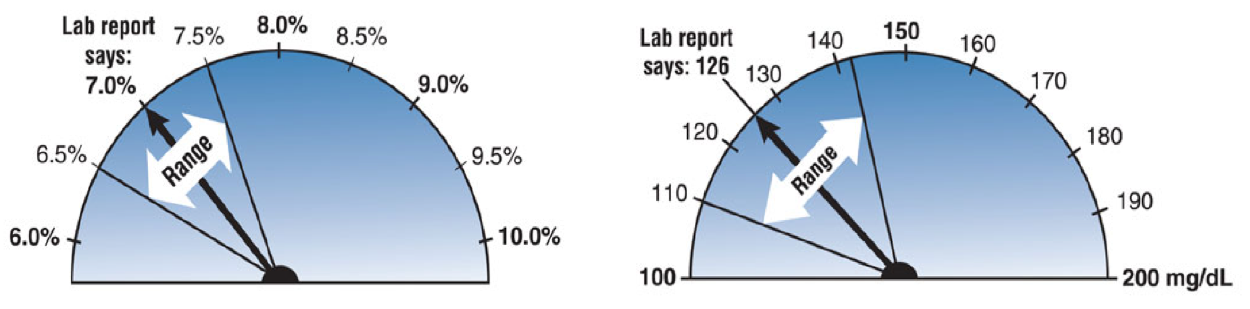
Using standardized IFCC methods, based on data generated to date, ELG falls within the acceptable tolerance
ELG-3 Corporation intends to submit a 510(k) for the ELG device as soon as the clinical data, as well as the complete manufacturing information is available. ELG expects it to be cleared for marketing as a Class 2 device. This is the likely regulatory pathway per previous submissions. We remain in close contact with the FDA during the clinical trial process.
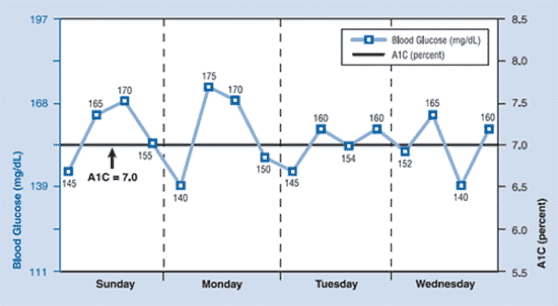
Currently we have 80 data points comparison to finger sticks fasting and non-fasting glucose OTC devices on the market. In addition, ELG shows CGMS “like” patterns, we will have more data from the 100 subjects with the intention of proving that ELG readings are consistent with CGMS.